|
4/11/2024 0 Comments Atom theory 
The diagram for 2 H subscript 2 shows two sets of bonded, white spheres. It is labeled one H subscript 2 molecule. Atoms themselves are composed of protons, neutrons, and electrons. 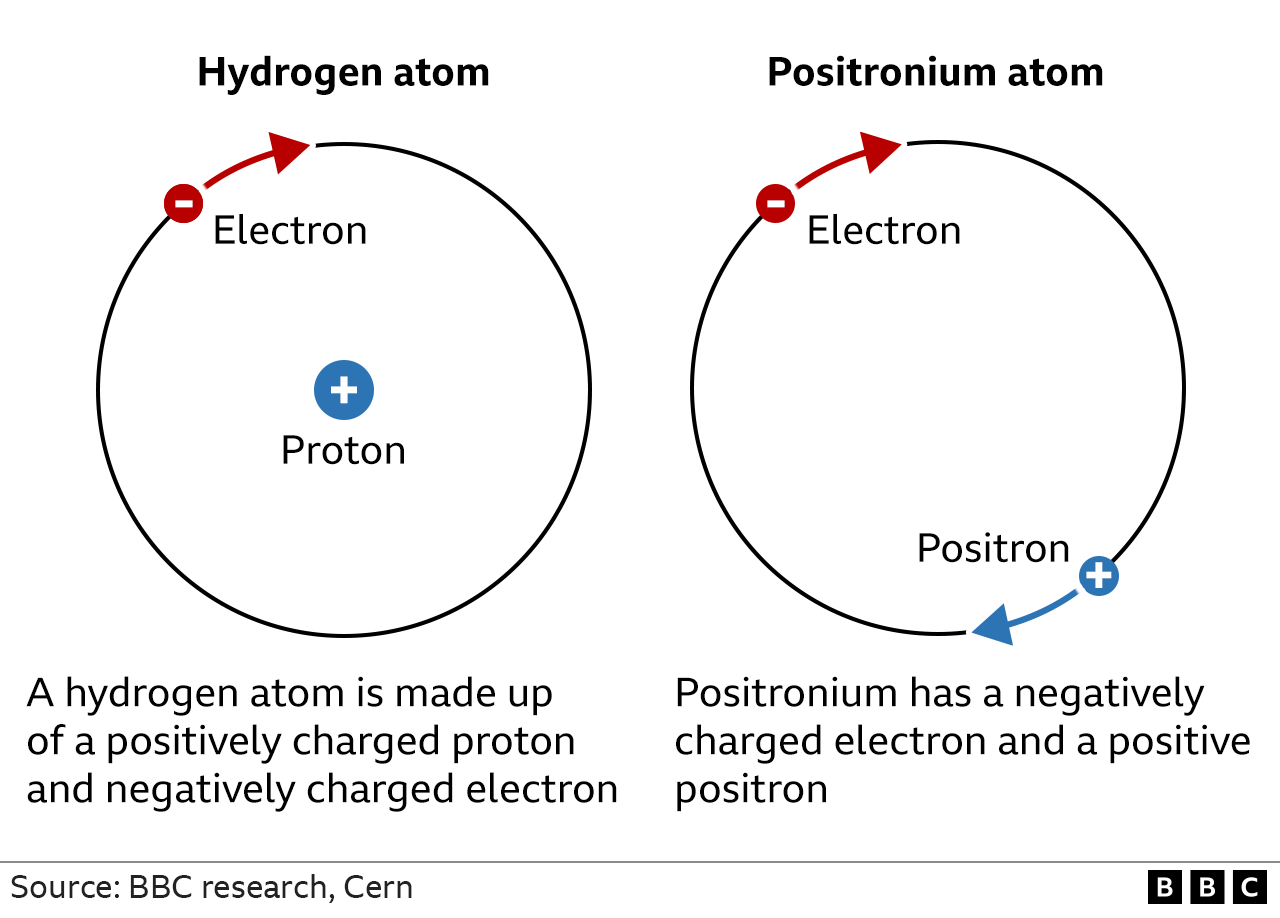
Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. Some said everything was made of water, which comes in three forms (solid ice, liquid water, and gaseous steam). 4.4: Atomic Theory is shared under a Public Domain license and was authored, remixed, and/or curated by LibreTexts. Beginning in about 600 BC, many Greek philosophers struggled to understand the nature of matter. The word ‘atom’ actually comes from Ancient Greek and roughly translates as. In fact, we have to go all the way back to Ancient Greece to find its genesis. 
Though our graphic starts in the 1800s, the idea of atoms was around long before. The diagram for H subscript 2 shows two white spheres bonded together. Atomic theory is the description of atoms, the smallest units of elements. This graphic takes a look at the key models proposed for the atom, and how they changed over time. The diagram for 2 H shows two white spheres that are not bonded together. The diagram for H shows a single, white sphere and is labeled one H atom. Atoms of different elements differ in size, mass, and other properties. Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed. The general tenets of this theory are: All matter is composed of extremely small particles called atoms. The law of multiple proportions states that if two elements form more than one compound between them, the masses of one element combined with a fixed mass of the second element form in ratios of small integers.\): The symbols H, 2H, H 2, and 2H 2 represent very different entities. Dalton proposed his atomic theory in 1804. Of course, we just as easily could have illustrated the law by considering the mass of nitrogen that combines with one gram of oxygen it works both ways!.Note that just as the law of multiple proportions says, the weight of oxygen that combines with unit weight of nitrogen work out to small integers there is a typo in Line 3-where there is a 3, there should be a 4. Protons and neutrons form the atomic nucleus. The three parts of the atom are protons (positively charged), neutrons (neutral charge), and electrons (negatively charged). By the late 1800’s, John Dalton’s view of atoms as the smallest particles that made up all matter had held sway for about 100 years, but that idea was about to be challenged. Line 3 is obtained by dividing the figures of the Line 2 by the smallest O:N ratio in Line 2 (which is the ratio for N 2O). The word ‘atom’ actually comes from Ancient Greek and roughly translates as ‘indivisible’. An atom is a building block of matter that cannot be broken apart using any chemical means. This module is an updated version of Atomic Theory I.Dalton based his theory on the law of conservation of mass and the law of constant composition. Atoms of the same element are the same atoms of different elements are different. Daltons atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties. It consists of three parts: All matter is composed of atoms. The published (1808) tenets of this theory were as follows: All matter is composed of extremely small particles called atoms. But someone who depends solely on experiment would work these out by finding the mass of O that combines with unit mass (1 g) of nitrogen. The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory, first stated by John Dalton, an English scientist, in 1808. In 1949, Maria Goeppert-Mayer one of only three women to have won the Nobel Prize in Physics and Hans Jensen independently proposed that the nucleus had a structure described by energy levels, similar to the theory behind how electrons are arranged in an atom. The numbers in Line 2 are just the mass ratios of O:N, found by dividing the corresponding ratios in line 1. At the same time, we probed even further into the nucleus. 
(These numbers were not known in the early days of Chemistry because atomic weights of most elements were not reliably known.) 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Each element has its own atomic number, which is equal to the number of protons in its nucleus. These ratios were calculated by simply taking the molar mass of each element, and multiplying by the number of atoms of that element per mole of the compound. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
